AI Takes Centre Stage in Pharma Policy Push at Sādhana Saptah 2026
Webinar spotlights next-gen drug discovery, faster approvals, and data-driven regulation New Delhi: The Department of Pharmaceuticals under the Ministry of Chemicals and Fertilizers on Saturday hosted a high-level webinar on
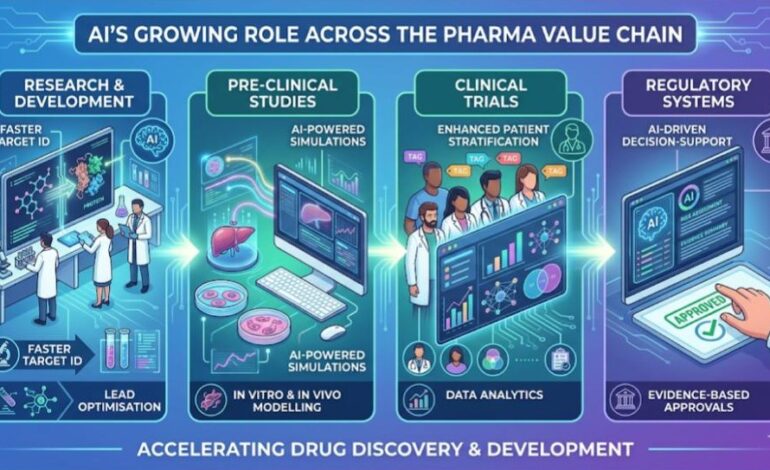
AI is transforming the pharmaceutical lifecycle by accelerating drug discovery, enhancing clinical trials through data-driven insights, and enabling smarter, evidence-based regulatory approvals.
Webinar spotlights next-gen drug discovery, faster approvals, and data-driven regulation
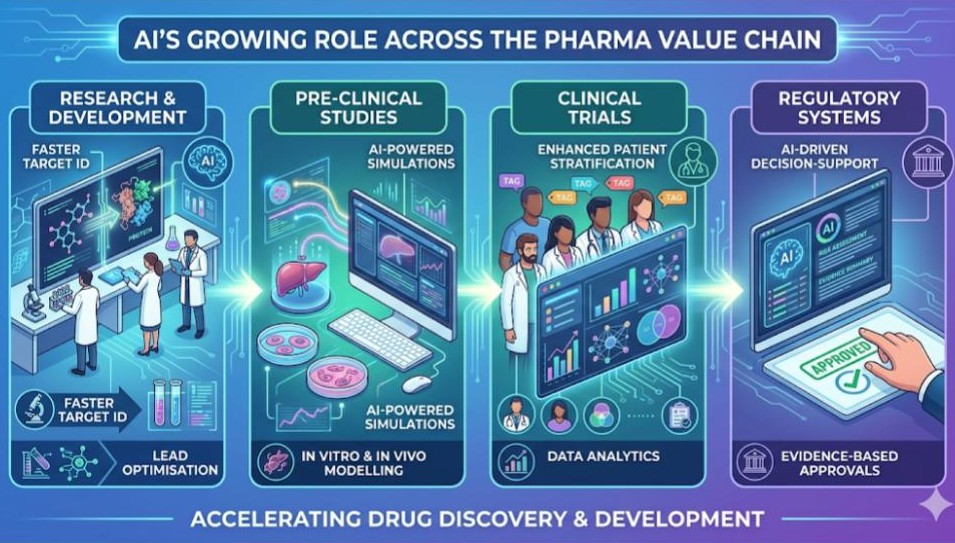
New Delhi: The Department of Pharmaceuticals under the Ministry of Chemicals and Fertilizers on Saturday hosted a high-level webinar on “AI and Emerging Technologies in Pharmaceuticals and Regulations,” positioning artificial intelligence as a critical force in reshaping India’s pharmaceutical ecosystem.
Organised as part of Sādhana Saptah 2026, the session underscored how AI is rapidly transforming the entire pharmaceutical lifecycle – from early-stage research to regulatory approvals and post-market surveillance.
Addressing officials and participants, Manoj Kumar, professor at the Department of Pharmacoinformatics, National Institute of Pharmaceutical Education and Research (NIPER), Mohali, highlighted how AI is accelerating innovation while reducing time and costs associated with drug development.
He outlined AI’s growing role across key stages of the pharma value chain. In research and development, AI is enabling faster target identification and lead optimisation. In pre-clinical studies, AI-powered simulations are improving in vitro and in vivo modelling. Clinical trials are benefiting from enhanced patient stratification and data analytics, while regulatory systems are increasingly adopting AI-driven decision-support tools for evidence-based approvals.
The session also explored cutting-edge advancements such as prediction of 3D protein structures, AI-led drug design, and de novo molecule generation. Emerging applications in polypharmacology and multi-target drug development were highlighted as promising areas for future breakthroughs.
Beyond discovery, AI is being leveraged in chemical synthesis, retrosynthetic pathway prediction, and drug repurposing. Tools that predict toxicity, bioactivity, and physicochemical properties are significantly improving early-stage research outcomes, enabling more efficient compound screening.
The webinar stressed the need for strengthening institutional capacity to fully harness AI’s potential in governance. This includes building capabilities for faster regulatory approvals, improving drug safety monitoring, and enhancing post-market surveillance systems.
The initiative aligns with the broader objectives of Mission Karmayogi, which aims to create a future-ready, citizen-centric civil service through continuous learning and digital enablement.
Sādhana Saptah, being held from April 2 to 8, is one of India’s largest collaborative capacity-building exercises. Spearheaded by the department of personnel and training, the capacity building commission, and karmayogi bharat, the programme brings together ministries, states, union territories, and training institutions to strengthen responsive and accountable governance under the theme “Ham Bane Karmayogi.”