Union Health Ministry Launches SHRESTH Index to Strengthen State Drug Regulatory Systems

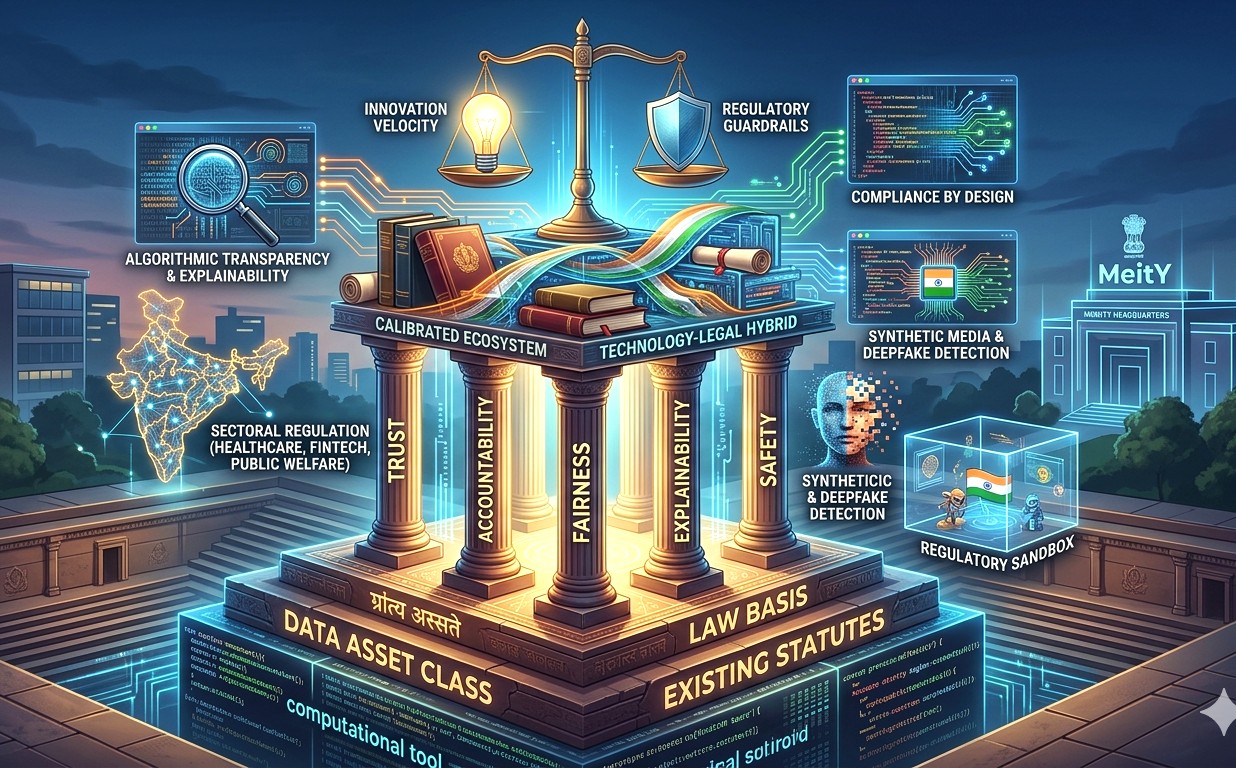
Union Health Secretary Punya Salila Srivastava launches the index virtually in the presence of Drug Controller General of India Dr Rajeev Singh Raghuvanshi in New Delhi on Tuesday
New Delhi, August 13: The Union Health Ministry on Tuesday launched the State Health Regulatory Excellence Index (SHRESTH), a first-of-its-kind national initiative designed to benchmark and strengthen state drug regulatory systems through a transparent, data-driven framework. The move aims to ensure consistent drug safety, quality, and efficacy standards across India.
Union Health Secretary Punya Salila Srivastava, who launched the index virtually in the presence of Drug Controller General of India Dr Rajeev Singh Raghuvanshi, described SHRESTH as a “virtual gap assessment tool” to help states evaluate their current capabilities and progress toward maturity certification.
The index, developed by the Central Drugs Standard Control Organization (CDSCO), will rank states in two categories — Manufacturing States and Primarily Distribution States/UTs — using 27 and 23 parameters respectively, across themes such as human resources, infrastructure, licensing, surveillance, and responsiveness. Data will be submitted monthly by states and scored by CDSCO to guide targeted improvements.
Srivastava urged stronger centre–state cooperation, emphasising that ensuring the quality of medicines is “a commitment of the Government to every home in India” and noting that India’s next goal is to elevate its medicines to the same global standard as its WHO ML3-certified vaccines.
The initiative also includes capacity-building workshops, expansion of the Not of Standard Quality (NSQ) Dashboard, and joint audits and trainings. State representatives welcomed the index, calling it a roadmap for harmonising regulatory processes and fostering innovation.
According to the Health Ministry, SHRESTH will drive cross-learning, promote uniform implementation of the Drugs and Cosmetics Act, and support the sharing of best practices to strengthen regulatory systems nationwide.